Global Medical Cannula Market to Reach USD 493.3 Million by 2036, Driven by Minimally Invasive Procedures and Emerging
DE, UNITED STATES, March 30, 2026 /EINPresswire.com/ -- In today’s fast-evolving healthcare landscape, clinicians and hospital leaders face constant pressure to deliver safer, more efficient procedures while managing rising patient volumes and chronic disease burdens. One quiet enabler gaining significant attention is the medical cannula — a simple yet critical device that supports everything from routine IV access to complex airway management and surgical interventions.
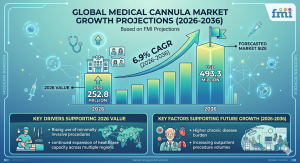
According to Future Market Insights (FMI), the global medical cannula industry is expected to be valued at USD 252.8 million in 2026. It is projected to grow at a steady 6.9% CAGR from 2026 to 2036, reaching USD 493.3 million by the end of the forecast period. This growth reflects broader trends: the expansion of minimally invasive techniques, increasing outpatient procedure volumes, and a clear shift toward advanced, patient-friendly designs that prioritize safety and ease of use.
Hospitals and surgical centers are actively investing in improved cannula technologies. Manufacturers are moving away from traditional metal formats toward biocompatible polymer-based options that offer better handling, reduced friction, and smoother insertion experiences. These upgrades — including refined tip designs, specialized coatings, and procedure-specific features — are helping clinicians achieve higher first-pass success rates and fewer complications, directly supporting better patient outcomes and workflow efficiency.
Quick Stats: Medical Cannula Market (2026–2036)
Industry Value in 2026: USD 252.8 Million
Forecast Value in 2036: USD 493.3 Million
Projected CAGR: 6.9%
Leading Product Segment: Guedel (27.8% share)
Key Growth Regions: India, China, Brazil
Prominent Players: AdvaCare Pharma, Teleflex Incorporated, B. Braun SE, ResMed Inc., Fisher & Paykel Healthcare Limited
Guedel cannulas continue to lead with a 27.8% share in 2026, thanks to their vital role in emergency airway management. Ongoing design improvements, such as better anatomical curvature, enhance patient comfort while maintaining reliability in high-pressure situations. Similarly, 18G gauge sizes command a strong 24.6% share, striking an ideal balance between optimal flow rates for rapid fluid or medication delivery and comfortable insertion — a critical factor in emergency departments and acute care settings.
Vascular access applications dominate the market with a commanding 74.2% share in 2026. This leadership stems from the universal need for reliable intravenous therapy, fluid management, blood sampling, and medication administration across hospitals. As interventional cardiology and catheter-based procedures expand, demand for specialized vascular cannulas with advanced insertion methods and performance-enhancing coatings continues to rise.
Regional Dynamics
Highlight Emerging Market Leadership Growth is particularly robust in emerging economies. India is projected to lead with an 8.2% CAGR, supported by rapid hospital expansion, government initiatives for healthcare modernization, and increasing domestic manufacturing of medical devices. China follows closely at 6.7% CAGR, driven by infrastructure investments and rising adoption of advanced surgical techniques. Brazil is expected to grow at 7.1% CAGR, fueled by urban surgical volume increases and improved device accessibility through public-private partnerships.
Even in mature markets, steady expansion continues. The United States is forecast to grow at 6.3% CAGR, where emphasis on precision, digital integration, and evidence-based protocols keeps innovation alive. Germany, known for its quality focus, is projected at 5.1% CAGR, with strong demand for high-performance devices aligned with rigorous safety and regulatory standards.
Market Drivers and Strategic Shifts
Several factors are accelerating uptake. Rising chronic disease prevalence is increasing the need for consistent vascular access. Hospitals are prioritizing infection prevention, leading to greater selection of cannulas with validated biocompatibility, appropriate gauge sizing, and coating technologies that minimize complications.
On the supply side, competition centers on clinical evidence and practical usability. Leading companies such as AdvaCare Pharma, Teleflex Incorporated, and B. Braun SE are investing in research collaborations, specialized product lines tailored to procedure complexity, and closer partnerships with healthcare providers. These strategies help align devices with real-world clinical protocols and training needs.
How the Market is Segmented
The medical cannula landscape is segmented by product type (Guedel, Yankauer, Berman, Frazier, and others), medical procedure (vascular access, surgery, intubation, suction, etc.), gauge size (14G to 26G), material (latex, latex-free, neoprene), and end user (hospitals, ambulatory surgical centers, clinics). This granular view helps stakeholders identify targeted opportunities — from flexible designs for minimally invasive work to rigid options suited for emergency use.
Request for Sample Report | Customize Report |purchase Full Report: https://www.futuremarketinsights.com/reports/sample/rep-gb-1057
Why Decision-Makers Should Explore Further
For procurement leaders, clinical directors, and strategic planners evaluating device portfolios, understanding these shifts is essential. The move toward polymer-based, procedure-specific cannulas is not just about compliance — it’s about making daily clinical work smoother, safer, and more predictable. As surgical volumes grow and infrastructure expands globally, suppliers who combine innovation with reliable distribution and clinical support are likely to gain stronger positioning.
Browse More Reports:
Preclinical Medical Device Testing Services Market: https://www.futuremarketinsights.com/reports/preclinical-medical-device-testing-services-market
Recycled Material Cleanroom Aura Respirators Market: https://www.futuremarketinsights.com/reports/recycled-material-cleanroom-aura-respirators-market
mRNA Cancer Vaccine Biologic Lines Market: https://www.futuremarketinsights.com/reports/mrna-cancer-vaccine-biologic-lines-market
Infant Medicine Syringes Market: https://www.futuremarketinsights.com/reports/infant-medicine-syringes-market
Infant Heart Monitor Market: https://www.futuremarketinsights.com/reports/infant-heart-monitor-market
About Future Market Insights (FMI)
Future Market Insights (FMI) is a leading provider of market intelligence and consulting services, serving clients in over 150 countries. Headquartered in Delaware, USA, with a global delivery center in India and offices in the UK and UAE, FMI delivers actionable insights to businesses across industries including automotive, technology, consumer products, manufacturing, energy, and chemicals.
An ESOMAR-certified research organization, FMI provides custom and syndicated market reports and consulting services, supporting both Fortune 1,000 companies and SMEs. Its team of 300+ experienced analysts ensures credible, data-driven insights to help clients navigate global markets and identify growth opportunities.
For Press & Corporate Inquiries
Rahul Singh
AVP - Marketing and Growth Strategy
Future Market Insights, Inc.
+91 8600020075
For Sales - sales@futuremarketinsights.com
For Media - Rahul.singh@futuremarketinsights.com
For web - https://www.futuremarketinsights.com
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.